![Find Local Clinical Studies in Los Angeles, CA [Trials] Study Travel Abroad | Explore Educational Trips & Global Learning Opportunities Find Local Clinical Studies in Los Angeles, CA [Trials] | Study Travel Abroad | Explore Educational Trips & Global Learning Opportunities](https://studyhardtravelsmart.com/wp-content/uploads/2026/05/th-88-300x200.jpg)
Research endeavors conducted within the Los Angeles metropolitan area, designed to evaluate the safety and efficacy of new medical treatments, interventions, or diagnostic procedures. These investigations, often involving human participants, adhere to... Read more »

A documented tool used to ensure the comprehensive and systematic preparation for a clinical research endeavor, addresses essential tasks and milestones before participant enrollment. This inventory ensures adherence to regulations, protocols, and... Read more »

The central question pertains to the efficacy of gold leaf applications on skin and whether scientific investigations substantiate any potential benefits. This exploration centers on rigorous, controlled experiments designed to evaluate objective... Read more »
![[Guide] Home Clinical Trial Visit by Nurse + Checklist Study Travel Abroad | Explore Educational Trips & Global Learning Opportunities [Guide] Home Clinical Trial Visit by Nurse + Checklist | Study Travel Abroad | Explore Educational Trips & Global Learning Opportunities](https://studyhardtravelsmart.com/wp-content/uploads/2026/03/th-265-300x200.jpg)
A structured document designed to provide standardized procedures and instructions for a healthcare professional, specifically a study nurse, when conducting a research-related visit to a participant’s residence. It outlines the necessary steps,... Read more »

The phrase identifies evaluations of medical interventions or products conducted by a specific organization. These assessments adhere to established scientific protocols and regulatory guidelines to determine the safety and efficacy of the... Read more »
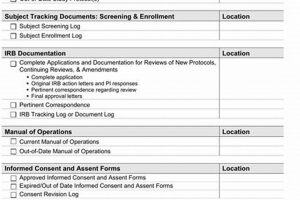
A structured tool designed to ensure study nurses are adequately prepared for the implementation of a novel research plan. It systematically verifies comprehension of the updated research procedures, data collection methods, ethical... Read more »

Research endeavors conducted within a specific metropolitan area in South Florida, involving human participants, aim to evaluate the safety and efficacy of novel medical interventions. These investigations may encompass pharmaceuticals, medical devices,... Read more »

Compensation for professionals overseeing the lifecycle of clinical trials is a multifaceted subject. This remuneration reflects experience, geographic location, employer type (pharmaceutical company, contract research organization, academic institution), and the complexity of... Read more »

A standardized document used by clinical trial study nurses during in-home patient visits facilitates the collection of consistent and accurate data. This tool often incorporates pre-defined fields for recording vital signs, medication... Read more »

Resources designed to aid individuals preparing for roles in managing clinical trials and related projects, available without cost, and structured in a checklist format for efficient study planning. These resources typically outline... Read more »


