
Investigations designed to assess the safety and efficacy of interventions aimed at improving individual or population health and well-being fall under a specific category. These endeavors adhere to rigorous scientific protocols and... Read more »
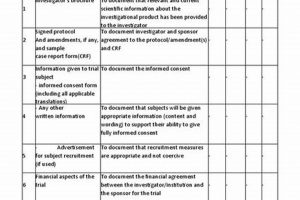
A structured tool aids a healthcare professional involved in research in executing a clinical investigation outside of traditional settings. It provides a systematic approach to ensure protocol adherence and patient safety when... Read more »
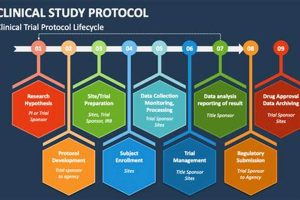
The systematic education of study nurses regarding the standardized guidelines and procedures governing clinical research endeavors is a critical component of ensuring data integrity and participant safety. This specialized instruction encompasses a... Read more »

A concise reference document covering essential information and procedures for the initial phase of a clinical research project is a valuable asset. This resource typically consolidates key regulatory guidelines, standard operating procedures,... Read more »


